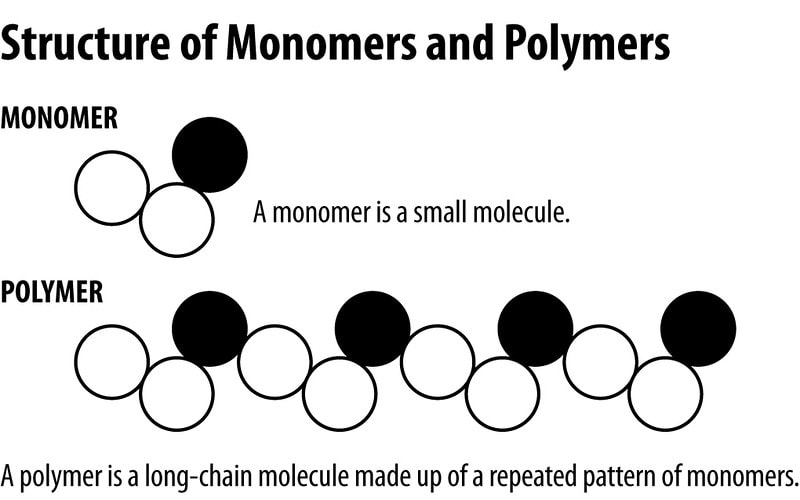
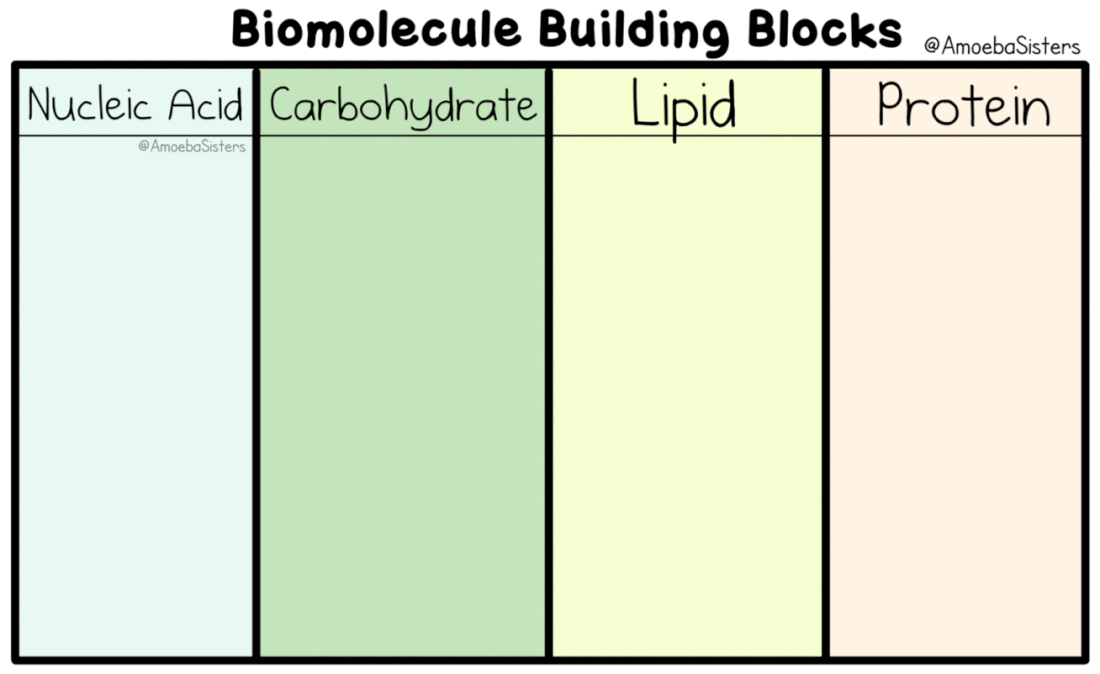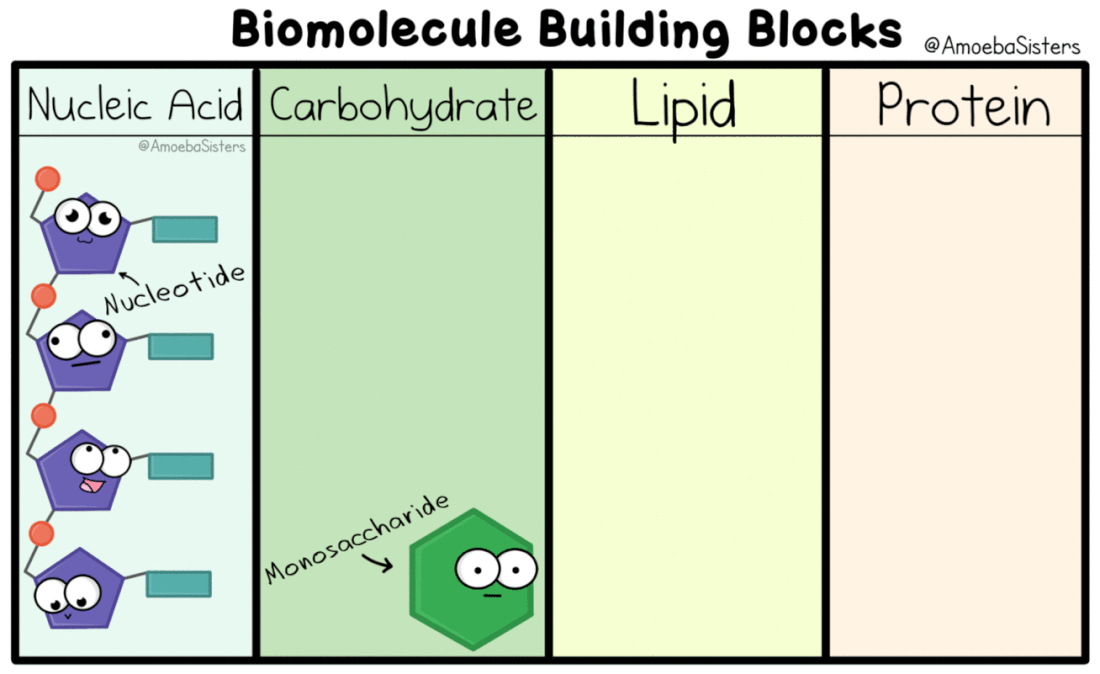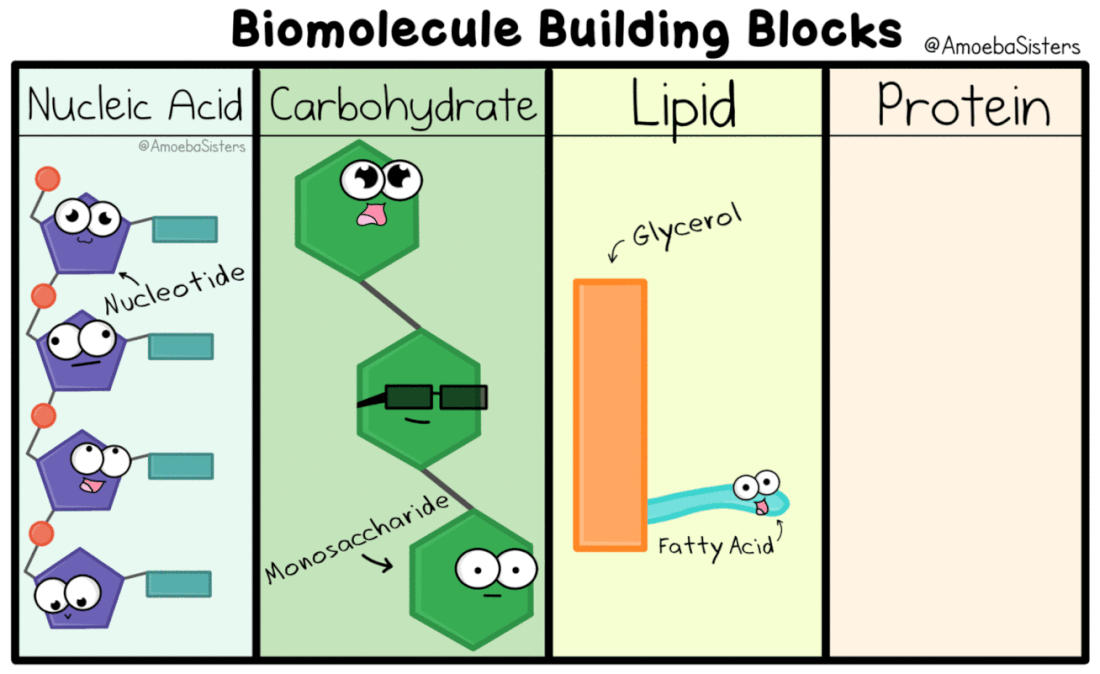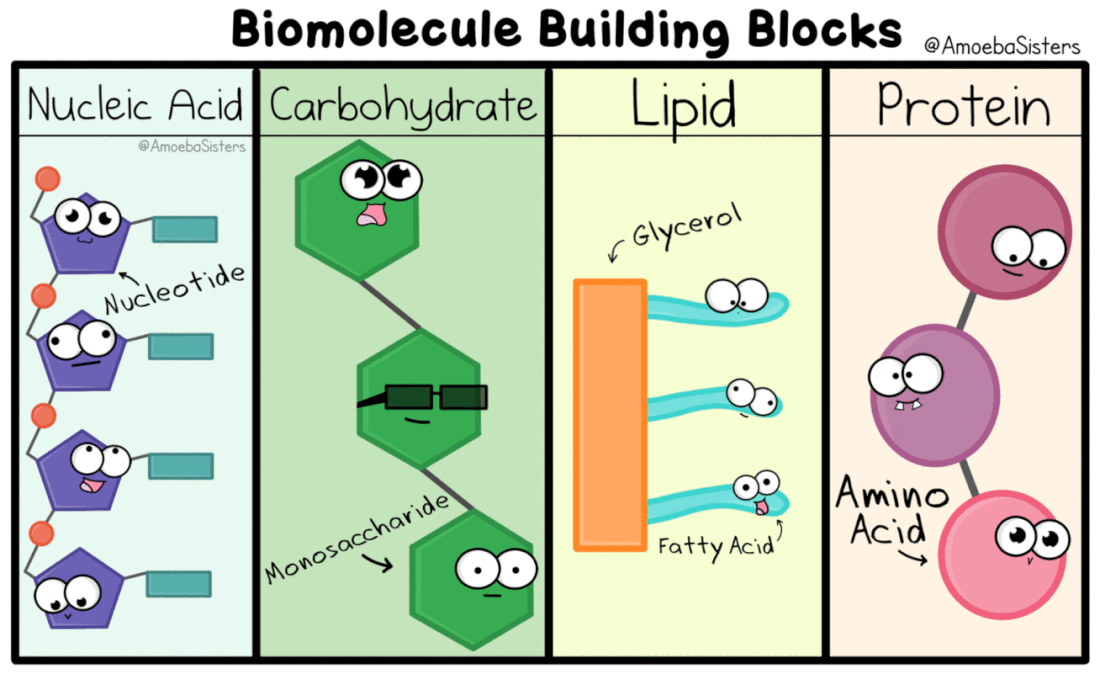
All living things are made of biomolecules. We get these biomolecules into our bodies through eating. The smallest part of each is called a monomer ("mono" means one). If I had a structure made of building blocks (like Legos) each block would be like a monomer of the structure. (watch this video)
Nucleic Acids
|
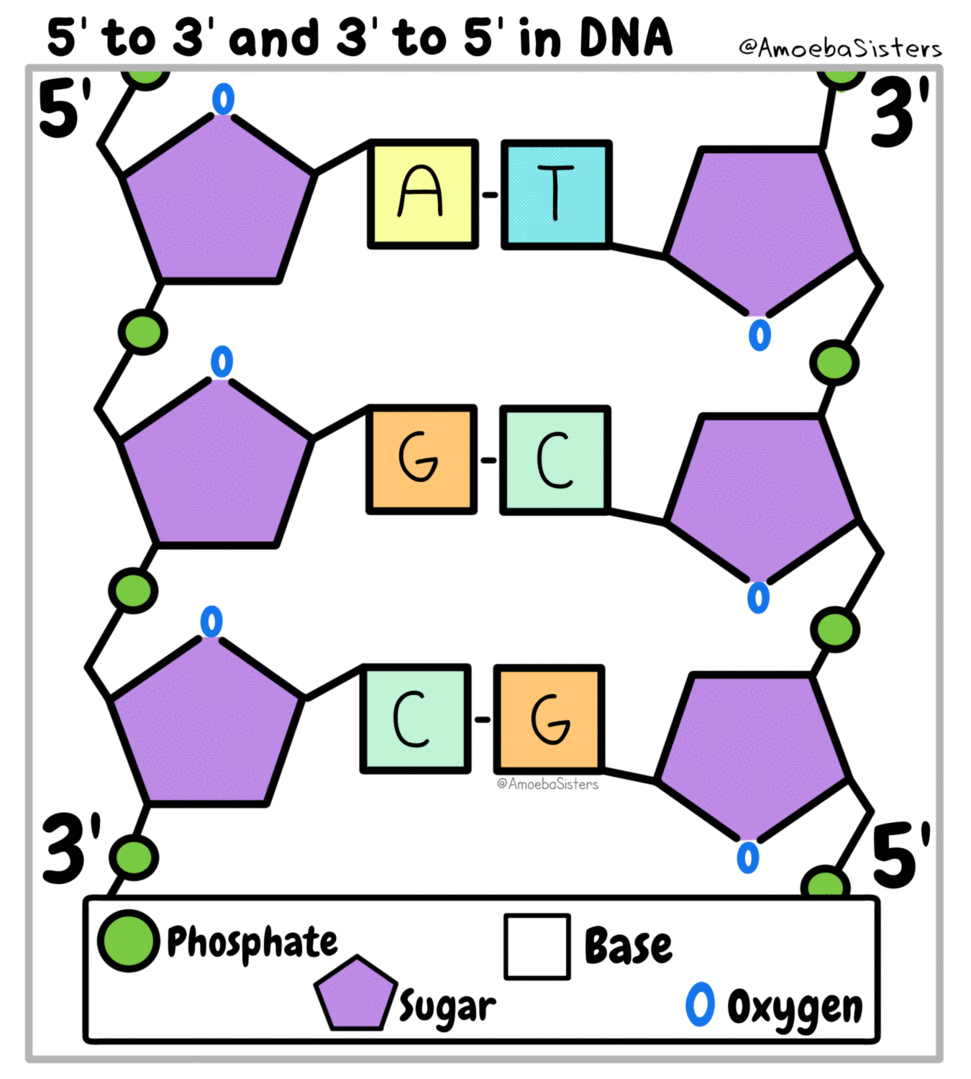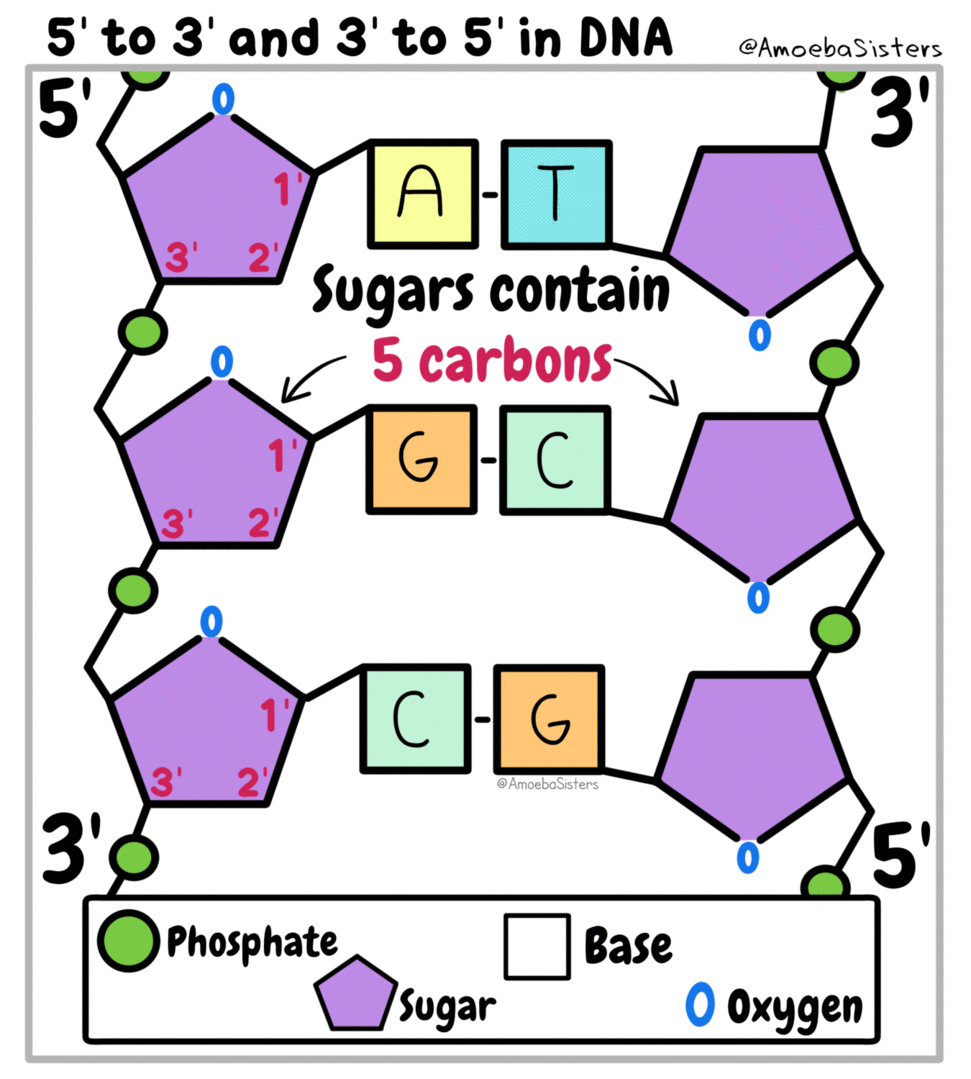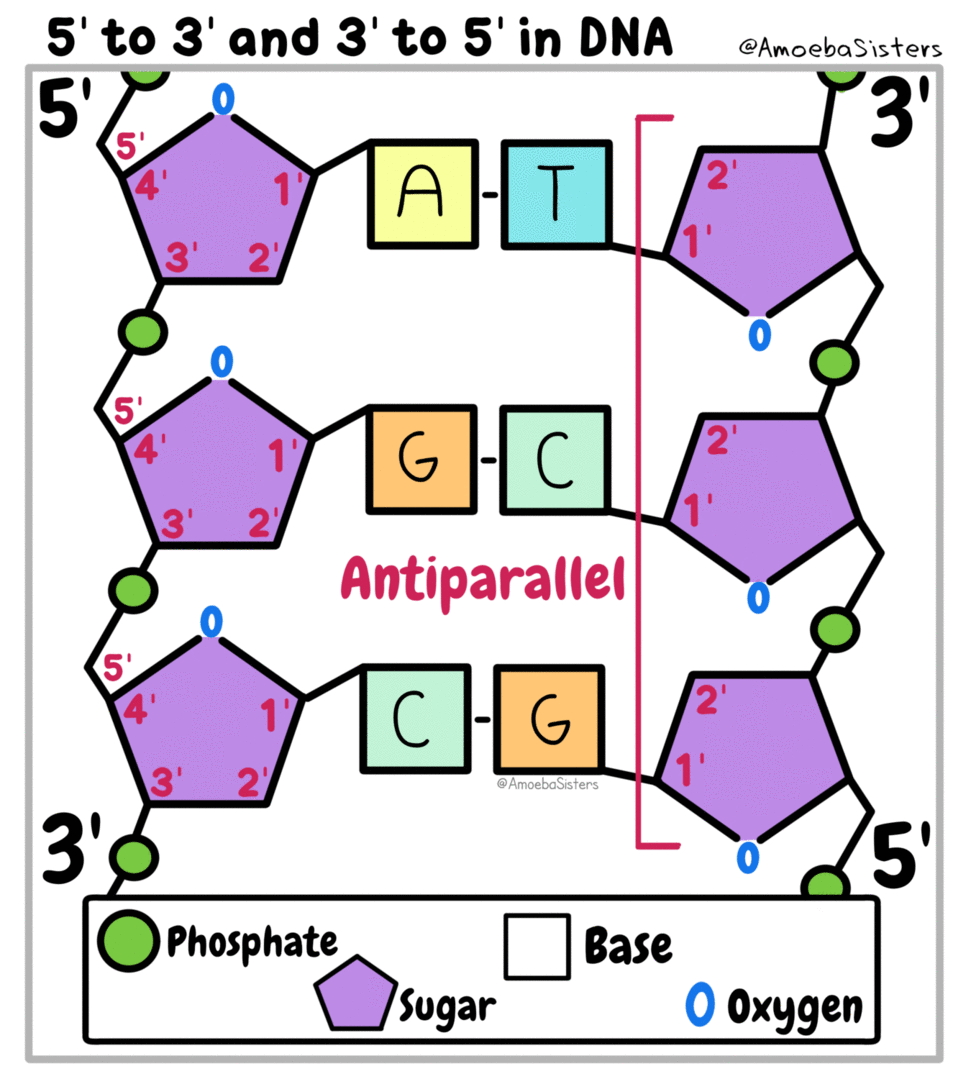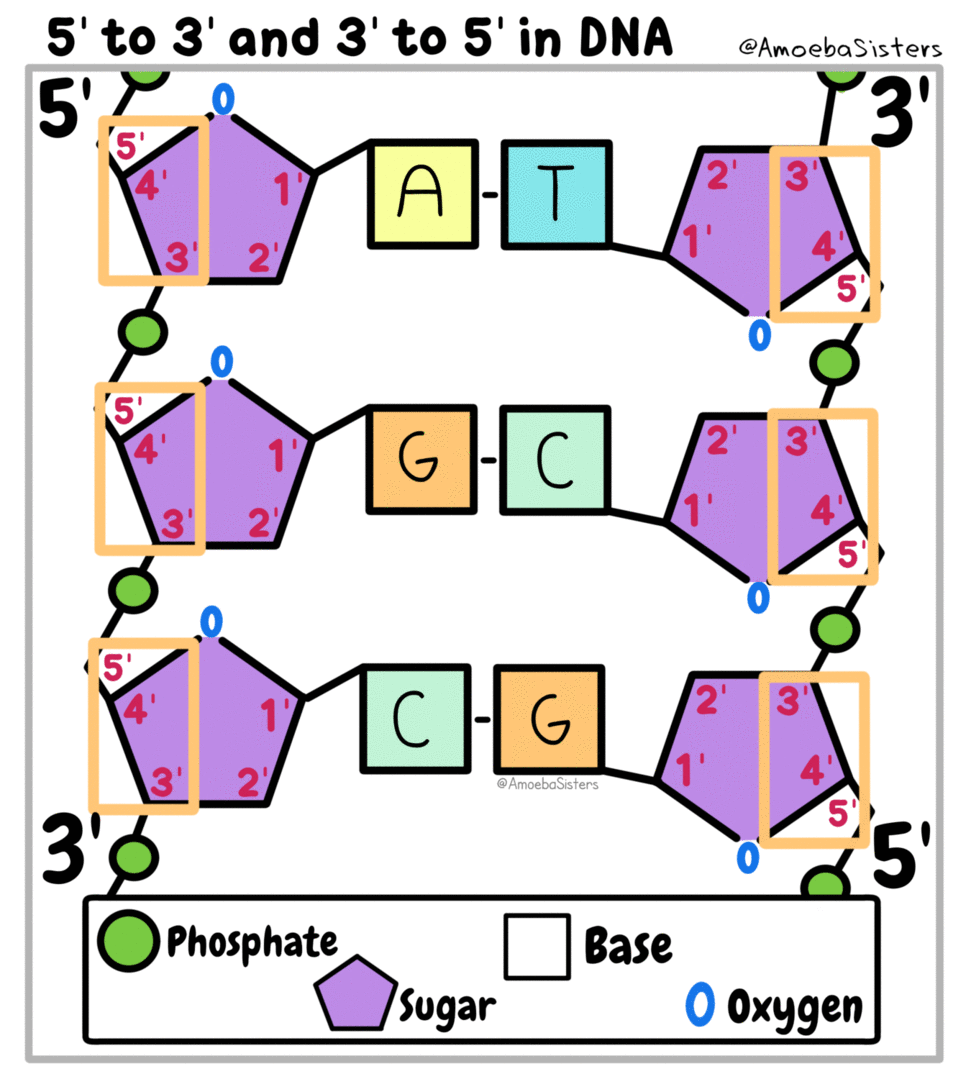
Genetic information, the information that tells our cells what to do, is kept in the form of nucleic acids. DeoxyriboNucleic Acid is kept in the nucleus of eukaryotic cells. RiboNucleic Acid can read the DNA and transfer the information to ribosomes that make proteins.
|
Carbohydrates
|
Carbo = Carbon
Hydrate = Water (Hydrogen and Oxygen) Carbohydrate = Carbon, Hydrogen, and Oxygen . Ratio = 1 : 2 : 1 Monomer = Monosaccharide Polymer = Polysaccharide |
Carbohydrates (or carbs) are very important sources of stored energy. They can be broken down quickly compared to other forms of stored energy, and that energy can be stored in ATP which, the cells can use to do work. If you play a sport that involves a lot of running, you may have a pasta party the night before an event. Pasta is heavy in carbohydrates, so the next day, your body will have made a lot of ATP in your sleep and will be able to continue this while you exert energy by breaking down the extra carbs in your system. Carbohydrates can also be found as receptors on the cell membrane and help make up structures like cell walls.
|
Lipids
|
*not technically monomers = Fatty Acid . + Glycerol
(Carbon, Hydrogen and Oxygen) |
In Biology, lipids, or fats, are used for long term energy storage. This is why it takes a lot of energy to burn fat in the body. You have fat on your body for insulation, it keep you warm. Lipids also make up the cell membrane and other cell structures.
|
Saturated vs Unsaturated
- In saturated fats, each carbon in the hydrocarbon chain is bound to two hydrogen atoms while in unsaturated fats, at least one carbon is bound to just one hydrogen.
- Saturated fats are easily packed together in the cell unlike the unsaturated fats.
- Saturated fats are solid at room temperature while unsaturated fats are liquid at room temperature.
Proteins
|
Monomer = Amino Acids
Carbon, Hydrogen, Oxygen, Nitrogen, (and sulfer) |
Proteins are use for a variety of cellular activities. They make up structures like muscles, they send and receive signal, they can be used for transport, the speed up reactions, and much more. There are 20 amino acids that are assembled in different combinations to make proteins.
|
Enzymes
Enzymes are a special type of protein that speed up reactions by lowering the activation energy, or the energy required to initiate a reaction. Every enzyme has an active site that is specific to a certain substrate. When the substrate attaches at the active site, the enzyme changes shape and the activation energy required for a reaction is lowered, allowing a reaction to occur. Once the reaction is over, the enzyme returns to its original shape and can be used by more of the same substrate.
Enzymes are a special type of protein that speed up reactions by lowering the activation energy, or the energy required to initiate a reaction. Every enzyme has an active site that is specific to a certain substrate. When the substrate attaches at the active site, the enzyme changes shape and the activation energy required for a reaction is lowered, allowing a reaction to occur. Once the reaction is over, the enzyme returns to its original shape and can be used by more of the same substrate.
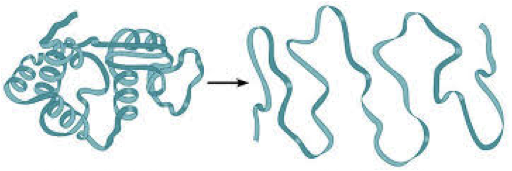
When a protein, like an enzyme, looses its shape it denatured. A denatured enzyme is non functional. Factors that can denature an enzyme are:
High Temperature Heavy metals, alcohols Low pH High pH
High Temperature Heavy metals, alcohols Low pH High pH